-
-
Hepatitis B Virus (HBV) Nucleic Acid Detection Kit (Fluorescence PCR Method)
KHB Hepatitis B Virus (HBV) Nucleic Acid Detection Kit is intended for quantitative detection of HBV DNA in human serum or plasma samples. This is used for assessment of response to antiviral therapy and monitoring of therapeutic effect. This kit is not used for blood screening.
Excellent performance on our automatic workstation PANA 9600S/X, PANA 3200S+ and all-in-one system Panall8000 .
-
-
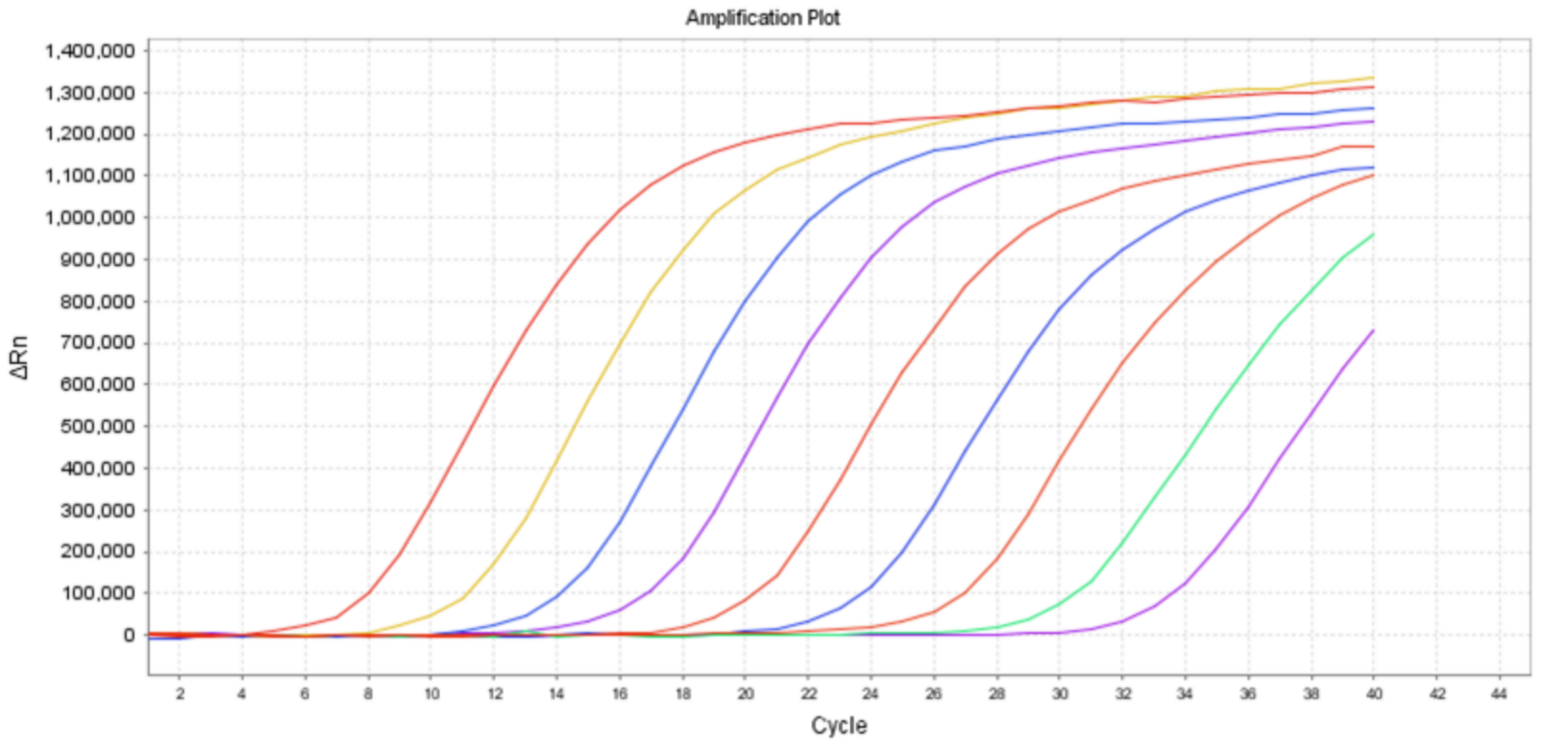
01Product Performance
High sensitivity: LoD: 5 IU/mL
Wide linear range:10~1.0×10^9 IU/mL
Broad genotype coverage: covering HBV genotypes A-H
Accurate quantification: internal standard-based quantification

.png)
-
02Clinical Significance
1.Accurately assess antiviral treatment response
2.Reliable monitoring of low-level viremia
3.Supports detection of occult HBV infection (OBI)
4.Early warning of HBV reactivation risk in immunosuppressed patients
5.Helps identify virological breakthrough during therapy

-
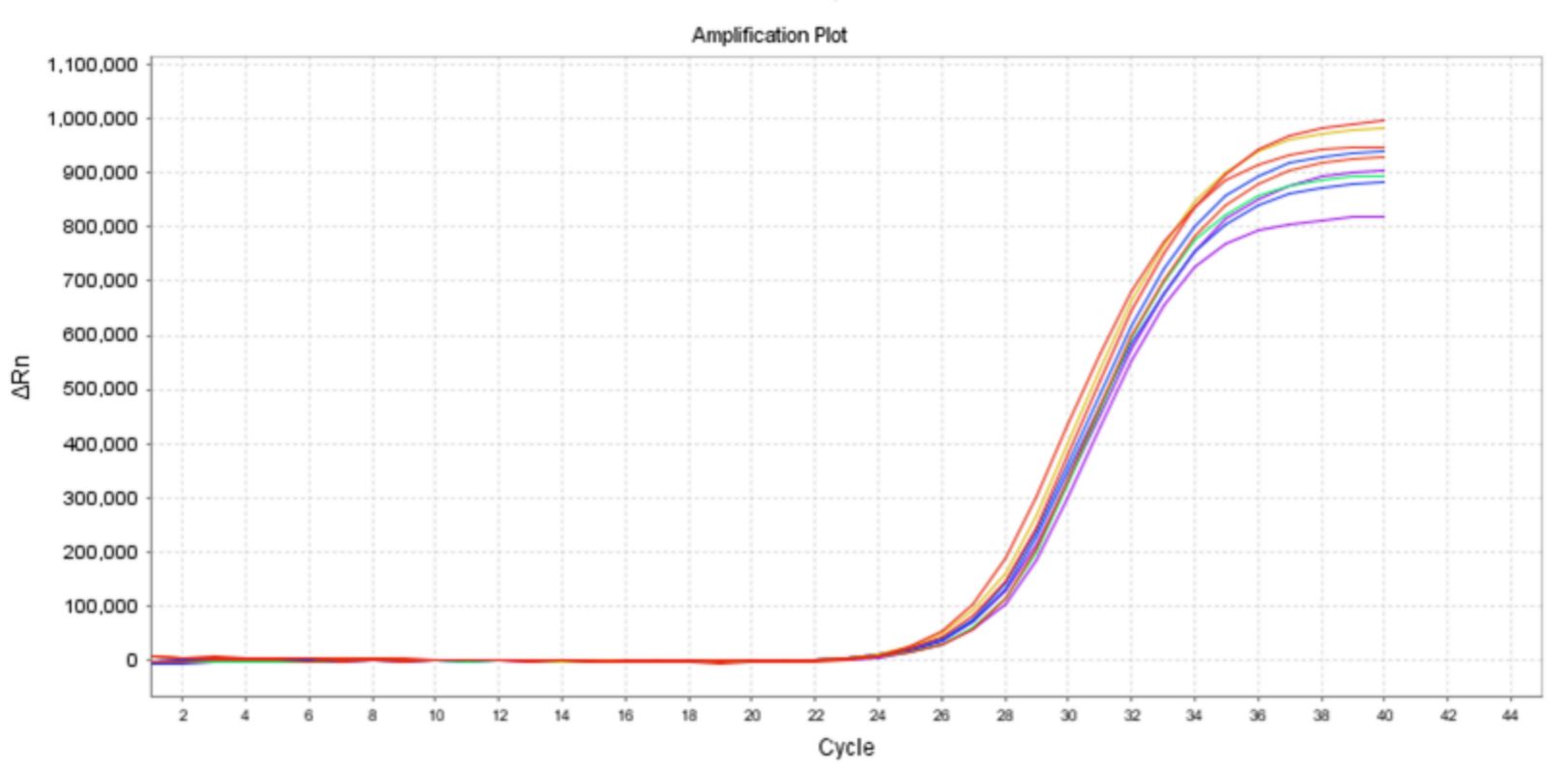
03Detection Principle
Internal standard quantification
Full-process internal control for reliable results
No run-specific standard curve required for simplified workflow
Minimize inter-tube variation and ensure batch reliability
Reduced contamination risk and reagent consumption
-807.png)
Contact us